Why is Diffusion Faster in Gases?
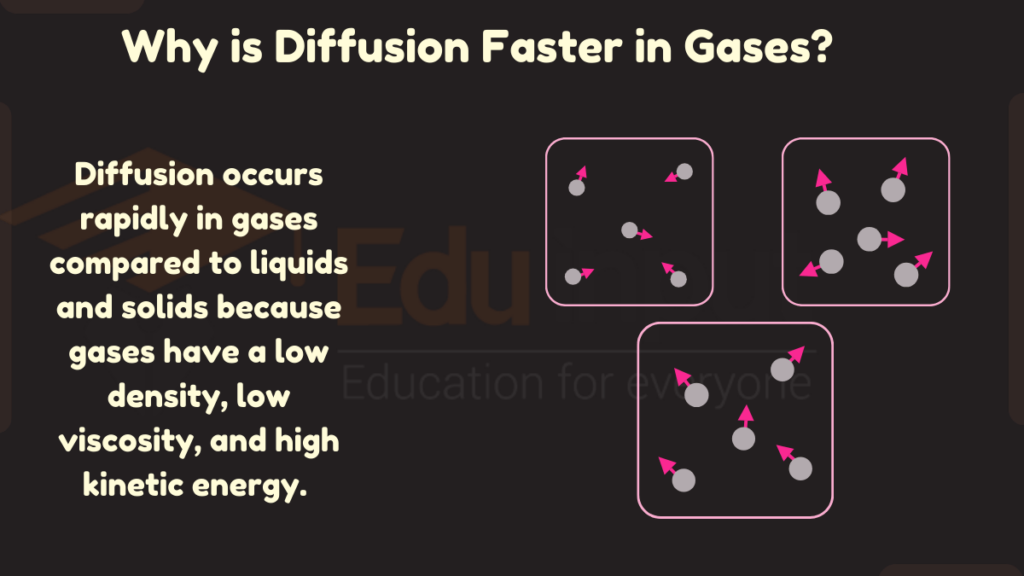
Diffusion occurs rapidly in gases compared to liquids and solids because gases have a low density, low viscosity, and high kinetic energy. The combination of low density, low viscosity, and high kinetic energy in gases allows for extremely fast diffusion rates.
Also learn about: Why Is Diffusion Faster In Water Than in Agar?
Reasons Why is Diffusion Faster in Gases
Here are few reasons Why Diffusion is Faster in Gases:
1. Low Density
Gases have low density because the molecules are spaced very far apart. This low density provides an open, unhindered environment for particles to diffuse through rapidly. With fewer gas molecules blocking the spaces in between, particles can migrate freely at high speeds.
2. Low Viscosity
Viscosity refers to a fluid’s resistance to flow. Gases have extremely low viscosity, meaning they offer very little resistance to particles trying to diffuse through them. With less thick fluid to slog through, particles can flow smoothly and quickly. This allows rapid dispersal and diffusion.
3. High Kinetic Energy
The molecules in gases move at very high speeds, giving them great kinetic energy. This kinetic energy gets transferred to particles diffusing through the gas. It speeds up their movement as well. The high-speed motion of gas molecules bombardment propels the diffusing particles forward faster.
Example
When spraying an air freshener, the scent molecules diffuse rapidly throughout the room because they can easily maneuver through the low-density, low-viscosity air. The high kinetic energy of the fast moving air molecules accelerates the dispersal. The same scent molecules would take much longer to diffuse through syrupy, dense liquid.




Leave a Reply