Molecular Orbital Theory, introduction, postulates, examples and applications
Introduction
Molecular Orbital Theory (MOT) is a fundamental concept in the field of chemistry, providing a comprehensive understanding of chemical bonding beyond the limitations of traditional theories. As we delve into the intricate world of molecules, this theory offers a nuanced perspective, unraveling the mysteries that simpler models fail to capture. In this article, we explore the fundamentals of Molecular Orbital Theory, shedding light on its significance in modern chemistry.
What is Molecular Orbital Theory (MOT)?
Molecular Orbital Theory is a quantum mechanical model used to describe the electronic structure of molecules. It involves the combination of atomic orbitals to form molecular orbitals, providing insights into bonding, stability, and reactivity
Limitations of Previous Bonding Theories
Before the advent of Molecular Orbital Theory, the Valence Bond Theory was a predominant model for understanding chemical bonding. However, it had its limitations, especially in explaining the electronic structure of complex molecules. Molecular Orbital Theory emerged as a more versatile approach, offering a unified framework to understand a broader range of molecular phenomena.
Postulates of Molecular Orbital Theory
- The overlapped atomic orbitals do not maintain their ientity.
- The overlapped atomic orbitals rearranged themselves to produce molecular orbitals.
- The number of molecular orbitals obtained is equal to the number of overlapped atomic orbitals.
- Half of the total number of molecular orbitals is called bonding molecular orbitals (BMO). Whereas the other half is called anti-bonding molecular orbitals (ABMO).
- Axial overlapping of two atomic orbitals belonging to two atoms results in the formation of a bonding molecular orbitals known as σ-orbital, and anti-bonding molecular orbital known as σ*-orbitals.
- Parallel overlapping of two atomic orbitals belonging to two toms results in the formation of a bonding molecular orbital known as π-orbital, and an anti-bonding molecular orbitals known as π*-orbital.
- A bonding molecular orbital is always lower in energy whereas anti-bonding molecular orbital is always higher in energy than either of the atomic orbitals of the two atoms.
- A covalent bond is formed when a bonding molecular orbital is filed by electrons leaving anti-bonding molecular orbital completely or partially empty.
- No covalent bond is formed when equal number of bonding and anti-bonding orbitals electrons are available.
- Filling of molecular orbitals takes place according to Auf-bau Principle. Pauli Exclusion Principle and Hund’s Rule.
- Number of covalent bonds formed between two atoms is called bond order. It can be calculated as
Bond Order = Number of electrons in BMO-Number of electrons in ABMO/2
Molecular Orbital Theory Examples
Hydrogen Molecule (H2)
In the case of a hydrogen molecule, two hydrogen atoms combine to form a diatomic molecule. Each hydrogen atom has a 1s atomic orbital. When they come together, these atomic orbitals combine to form two molecular orbitals: a bonding molecular orbital (σ) and an antibonding molecular orbital (σ*).
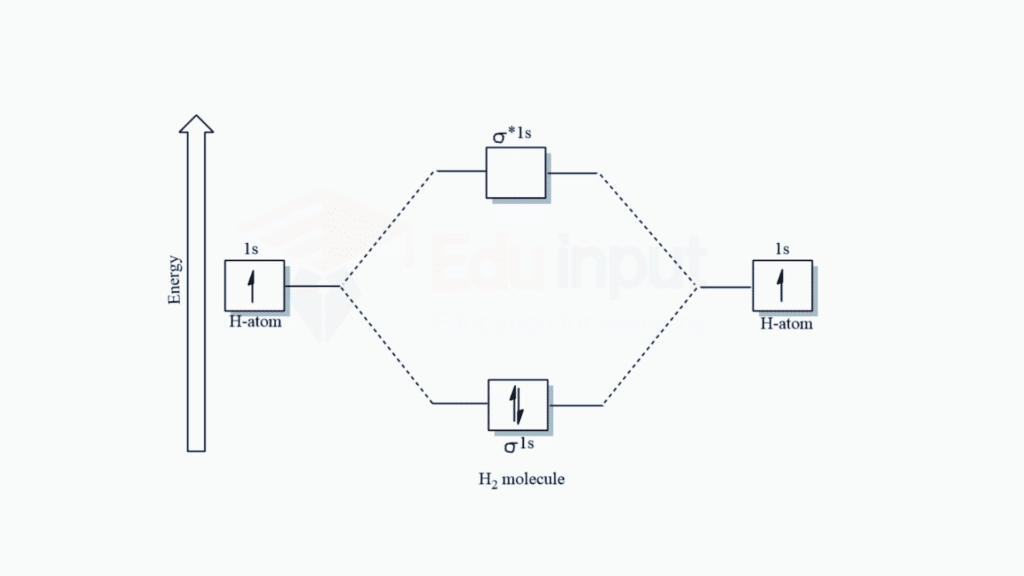
Bonding Molecular Orbital (σ)
The two 1s atomic orbitals overlap constructively, leading to the formation of a bonding molecular orbital. Electrons in this orbital contribute to the stability of the molecule.
Antibonding Molecular Orbital (σ)*
The two 1s atomic orbitals overlap destructively, forming an antibonding molecular orbital. Electrons in this orbital would destabilize the molecule.
Oxygen Molecule (O2)
Oxygen has six valence electrons (2s² 2p⁴). In the case of O2, two oxygen atoms combine. The molecular orbital diagram for oxygen involves the combination of 2s and 2p orbitals.
Sigma (σ) and Pi (π) Bonds: Oxygen molecules form a sigma (σ) bond by head-to-head overlap of two atomic orbitals and two pi (π) bonds by side-to-side overlap.
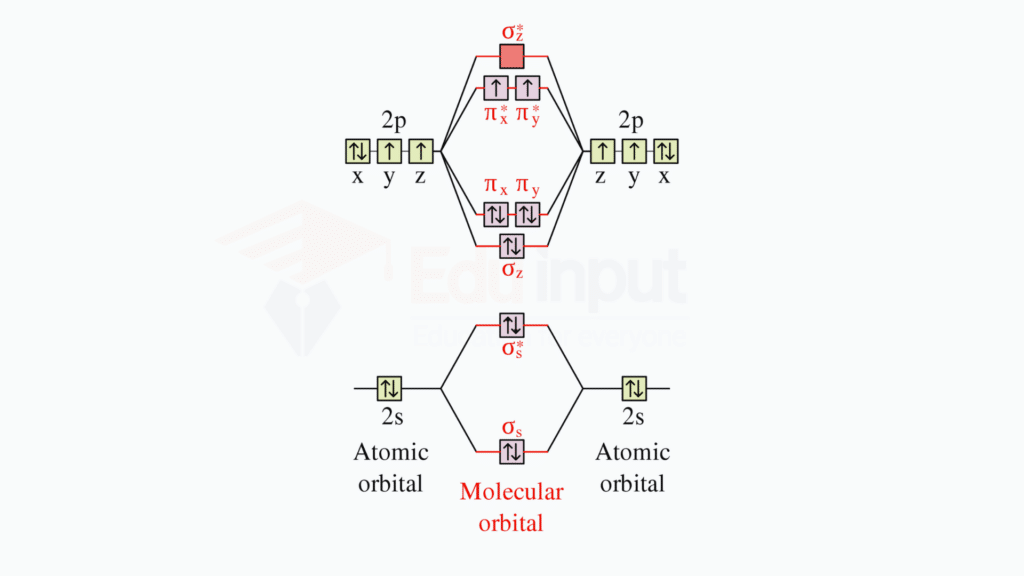
σ Bonding Molecular Orbital
The overlap of the two 2p orbitals along the internuclear axis forms a σ bonding molecular orbital.
π Bonding Molecular Orbitals
The sideways overlap of the two sets of 2p orbitals results in two π bonding molecular orbitals.
σ and π Antibonding Molecular Orbitals**
There are corresponding σ* and π* antibonding molecular orbitals formed by the destructive overlap of atomic orbitals.
Nitrogen Molecule (N2):
Atomic Orbitals of Nitrogen: Nitrogen has five valence electrons, and its electron configuration is 2s2, 2p3. In the molecular orbital theory, the 2s and 2p orbitals participate in the formation of molecular orbitals.
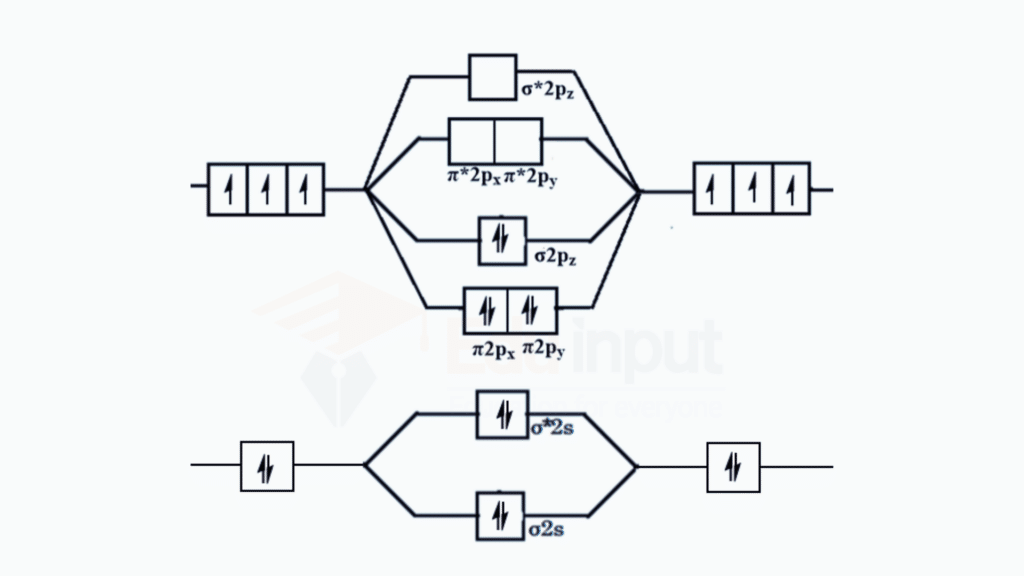
Formation of Sigma (σ) and Pi (π) Bonds
σ Bonding Molecular Orbital: The head-to-head overlap of two 2p orbitals along the internuclear axis results in a σ bonding molecular orbital. This is the primary bond in the nitrogen molecule.
π Bonding Molecular Orbitals
Side-to-side overlap of the remaining 2p orbitals results in two π bonding molecular orbitals.
σ and π Antibonding Molecular Orbitals**
Corresponding σ* and π* antibonding molecular orbitals are formed by the destructive overlap of atomic orbitals. The nitrogen molecule is often represented with a triple bond, consisting of one σ bond and two π bonds. The σ bond is formed by the overlap of the two 2p orbitals along the internuclear axis. The two remaining 2p orbitals overlap sideways to form the two π bonds.
Key Concepts of Molecular Orbital Theory
At the heart of Molecular Orbital Theory lies the concept of molecular orbitals – regions of space where electrons are likely to be found in a molecule. These orbitals result from the constructive and destructive interference of atomic orbitals as atoms come together to form molecules. The theory introduces the terms bonding, antibonding, and nonbonding molecular orbitals, each playing a crucial role in determining the stability and properties of molecules.
Atomic Orbitals
To comprehend Molecular Orbital Theory, a solid understanding of atomic orbitals is imperative. We revisit the basic types – s, p, d, and f – and delve into their unique shapes. The Aufbau principle and Hund’s rules, governing the filling of electrons in atomic orbitals, provide insights into the electronic configuration of atoms. Furthermore, we explore the correlation between atomic orbitals and electron energy levels, setting the stage for the transition to molecular orbitals.
Molecular Orbitals
The amalgamation of atomic orbitals leads to the creation of molecular orbitals. Understanding the different types – bonding, antibonding, and nonbonding – is crucial for predicting the behavior of molecules. Molecular orbitals, depicted through diagrams, offer a visual representation of the distribution of electrons in a molecule. This section explores molecular orbital diagrams for simple molecules like H2, O2, and N2, unraveling the intricate dance of electrons in the formation of chemical bonds.
Bonding and Antibonding Molecular Orbitals
The energy and shape of molecular orbitals are influenced by various factors. This section dissects these influences and explains how bonding molecular orbitals contribute to the stability of molecules, while antibonding molecular orbitals have a destabilizing effect. Molecular orbital diagrams are powerful tools that enable us to predict bond order, bond length, and magnetic properties, providing a deeper understanding of molecular structures.
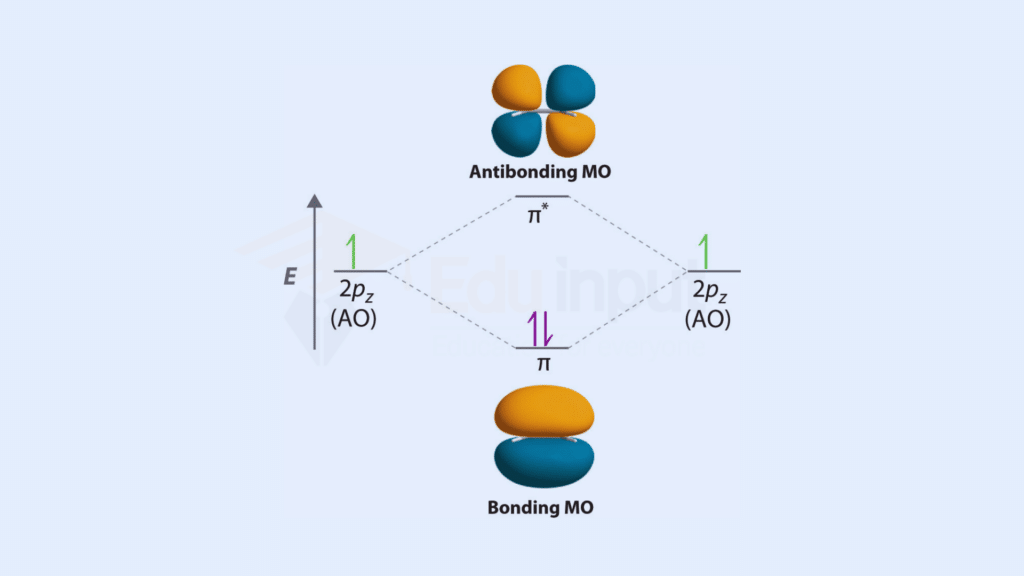
Also read: Difference between Bonding and Antibonding Molecular Orbitals
Applications of Molecular Orbital Theory
Molecular Orbital Theory (MOT) is a fundamental concept in quantum chemistry that provides insights into the electronic structure and properties of molecules. The theory is based on the idea of molecular orbitals, which are formed by the combination of atomic orbitals. Molecular Orbital Theory finds wide applications in various fields. Here are some key applications:
Molecular Stability and Reactivity
MOT is crucial for predicting the stability and reactivity of molecules. It helps in understanding how molecular orbitals contribute to the overall stability of a molecule and how the arrangement of electrons influences its reactivity in chemical reactions.
Bonding in Diatomic Molecules
MOT is extensively used to describe the bonding in diatomic molecules. It explains the formation of sigma (σ) and pi (π) bonds and provides a quantitative understanding of bond strength and bond order.
Polyatomic Molecules
MOT can be applied to polyatomic molecules, providing insights into their electronic structure. It helps in predicting the geometry and stability of molecules with multiple atoms.
Conjugated Systems in Organic Chemistry
In organic chemistry, MOT is applied to understand conjugated systems, such as in aromatic compounds. It explains the delocalization of π electrons and helps predict the stability and properties of aromatic molecules like benzene.
Transition Metal Complexes
Molecular Orbital Theory is widely used to explain the electronic structure and bonding in transition metal complexes. It helps understand the color, magnetic properties, and reactivity of these complexes.
Predicting Electronic Spectra
MOT is employed to predict electronic spectra, especially in the ultraviolet (UV) and visible regions. It explains the absorption of light by molecules and is crucial in fields like spectroscopy.
Chemical Bonding in Coordination Compounds
Coordination compounds often involve complex bonding situations. MOT is used to understand the nature of bonding in these compounds, including the formation of metal-ligand bonds.
Prediction of Magnetic Properties
MOT is instrumental in predicting the magnetic properties of molecules. It helps explain the presence of magnetic moments in certain compounds based on the arrangement of electrons in molecular orbitals.
Understanding Photochemical Reactions
MOT is applied to understand photochemical reactions, particularly those involving the absorption of light by molecules. It provides insights into the electronic transitions that occur during photochemical processes.
How does MOT differ from Valence Bond Theory (VBT)?
MOT considers the formation of molecular orbitals from the combination of atomic orbitals, allowing for delocalization of electrons. Valence Bond Theory, on the other hand, describes bonding as the overlap of atomic orbitals, emphasizing localized electron pairs.
What are Molecular Orbitals?
Molecular orbitals are mathematical functions that describe the distribution of electrons in a molecule. They result from the combination of atomic orbitals and define the regions in a molecule where electrons are likely to be found.
How does MOT explain bonding in molecules?
MOT explains bonding by showing the constructive interference of atomic orbitals, leading to the formation of bonding molecular orbitals. It also highlights the destructive interference, resulting in the creation of anti-bonding molecular orbitals.
What is the significance of Bond Order in MOT?
Bond order, derived from the difference between the number of bonding and anti-bonding electrons, indicates the strength and stability of a bond. Higher bond orders generally correspond to stronger and shorter bonds.
How does MOT apply to diatomic molecules?
Answer: In diatomic molecules, MOT explains the formation of sigma (σ) and pi (π) bonds. It helps predict the electronic structure, bond strength, and overall stability of these molecules.
Can MOT predict the magnetic properties of molecules?
Yes, MOT is applied to predict the magnetic properties of molecules by analyzing the arrangement of electrons in molecular orbitals. It helps explain the presence of magnetic moments in certain compounds.
Can MOT be applied to polyatomic molecules?
Yes, MOT applies to polyatomic molecules. It provides insights into the electronic structure, stability, and reactivity of molecules with multiple atoms.




Leave a Reply