Density-Definition, Formula, Examples, And Application
Measurement of density compares the amount of matter an object has to its volume. An object with a lot of matter in a small volume has a high density. We will learn about the units of density and the definition of density in this article.
What is Density?
The density is the same for a pure substance as it is for a mass concentration. Osmium and iridium are the densest known elements at standard conditions for temperature and pressure, and they are relevant to buoyancy, purity, and packaging.
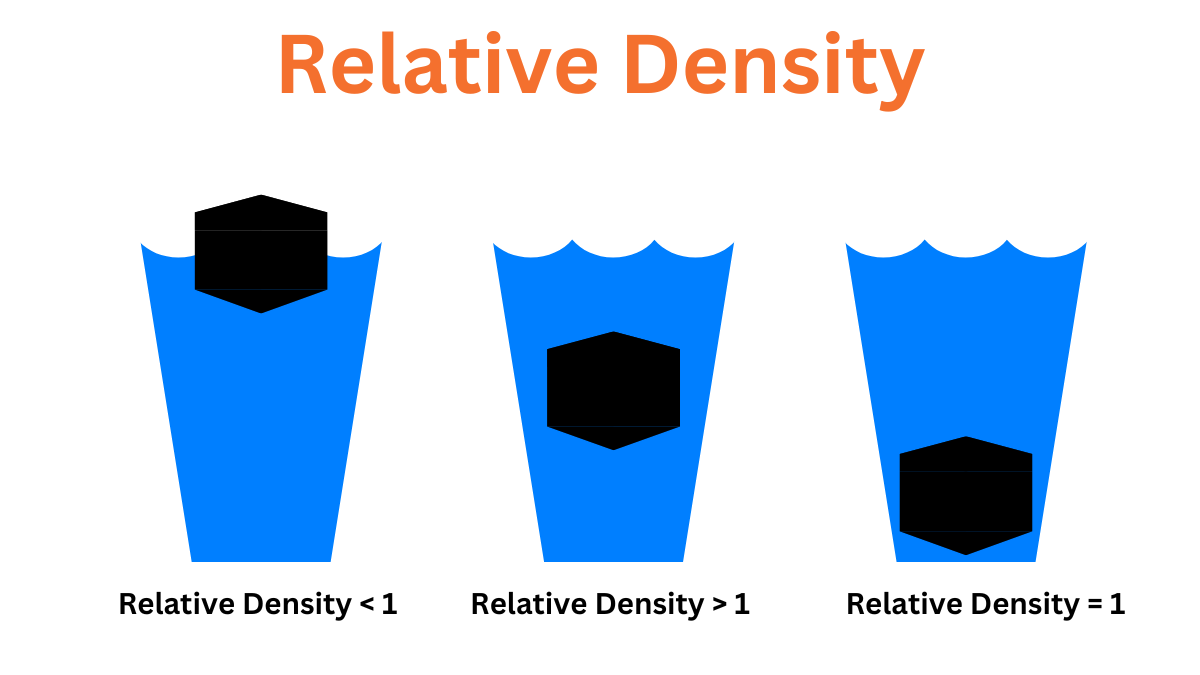
In order to simplify comparisons of density across different systems of units, it is sometimes replaced by the dimensionless quantity “relative density” or “specific gravity”.
Water is usually the ratio of the density of the material to that of a standard material. A relative density of less than one means that the substance floats in the water. The density of a material can change depending on pressure and temperature.
This variation is much larger for gases than it is for liquids. Increasing the pressure on an object decreases the object’s volume and increases its density.
Increasing the temperature of a substance decreases its density by increasing its volume. When heating the bottom of a fluid, the heat from the bottom to the top causes it to rise in density, which is related to the decrease in the density of the heated fluid.
Sometimes referred to as its specific volume, the density of a substance’s specific volume is called the reciprocal of the density. Increasing the amount of a substance does not increase its density, rather it increases its mass.
Formula of density
Where ρ is density, M is mass, and V is volume is where the formula for density is.
ρ=M/V
Units of Density
The units of grams per cubic centimeter are used to express density. Water has a density of 1 gram per centimeter, while Earth has a density of 5.51 grams per centimeter. In metre-kilogram-second or SI units, the density can be expressed as kilograms per cubic meter.
The density of air is 1.2 kilograms per square meter. The mass of a body is equal to the volume divided by the density, while the volume is equal to the mass divided by the density.
There are some more units of density.
| gram per millilitre (g/mL) | metric ton per cubic meter (t/m3) |
| kilogram per litre (kg/L) | megagram (metric ton) per cubic meter (mg/m3) |
| kilogram per cubic decimetre (kg/dm3) | gram per cubic centimeter (g/cm3) 1 g/cm3 = 1000 kg/m3 |
Density Examples
Examples of dense materials include iron, Platinum, and lead. A lot of types of rock and minerals are examples of dense material. The materials that are dense are more likely to be heavy or hard. The opposite of dense is sparse and a few examples of sparse materials are glass, bamboo, aluminum, and styrofoam.
Liquids are less dense than Solids and gases are less dense than Liquids. Solids, liquids, and gases all have particles that are free to move all over the place due to the fact that they all have densely packed particles.
Applications of Density in Real Life
The densities of two substances help you in separation techniques, for example, in pipe design, shipbuilding, helium balloons, weight distribution in the airplane, and the fact that ice floats on water.
Oil drops start to float on the water due to less density than the water if there is a leak in an oil tank in the ocean. Density is used to determine whether an object will float on the water or not. The density difference is the reason for the floating of ships and the diving of submarines.
.






Leave a Reply