Solid State Physics-The Science Behind The Physics of Solid Objects
Solid State Physics is all about the properties of solids. The concept of solids is very much different when compared to fluids. Fluid is liquid or a substance that flows. Whereas, solid is a non-flowable state of matter.
Solid State Physics
Solid consists of atoms or molecules that are joined together to form an ordered structure. Solids are the building blocks of all living creatures. We can say that the solid state is the foundation of the entire universe.
Metals are the hardest substances in the world. They resist any kind of deformation. They are mostly used for making alloys and for manufacturing the machines like aircraft.
Metals consist of a very large number of electrons that are arranged in the outermost layer of the metal. When you touch a metallic object, you will feel cold because the metal will give out a lot of energy to you.
Basic Properties of Solids
A solid is an object that has some basic properties that are given below.
Density
Density is the measure of the amount of matter within a specific volume of space.
Hardness
Hardness is the resistance that an object offers against being deformed. A hard material resists being compressed, whereas a soft material yields easily.
Elasticity
Elasticity is the property of a material in which it can recover its original shape after it is deformed. A soft solid is easily deformable, and it may return to its original shape after being deformed. A hard solid is less likely to yield.
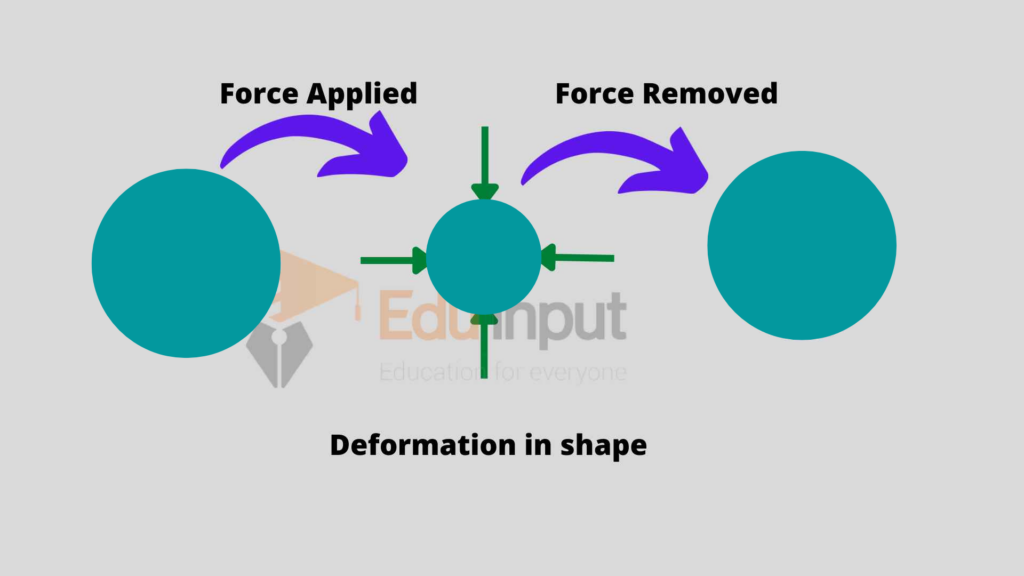
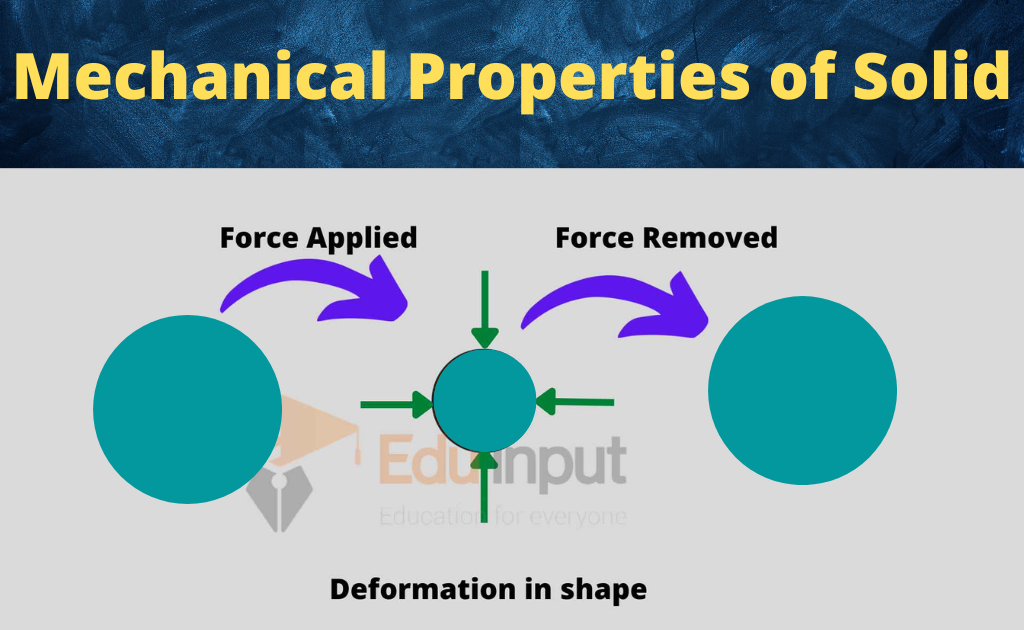
Mechanical properties
Mechanical properties of solids include deformation in shape, stress, strain, elastic constant, and yield strength.
Electronic Properties
Solid-state physics properties such as electrical conduction and heat capacity are investigated. The Drude model was one of the first approaches to electron transport in solid.
It is a simplified version of the Drude-Lorentz equation, an attempt to explain the electrical charge by the electrons.
The material was thought to have an immobile positive ion and an electron gas of classical, non-Interacting electrons. The Drude model underestimated the electronic heat capacity, despite being able to explain electrical and thermal Conductivity and the Hall effect.
Electrical properties
Electrical properties of solids include energy band theory and intrinsic and extrinsic semiconductors. The fundamental property of solids is their ability to conduct electric current.
There are three types of solids on the basis of conduction which are
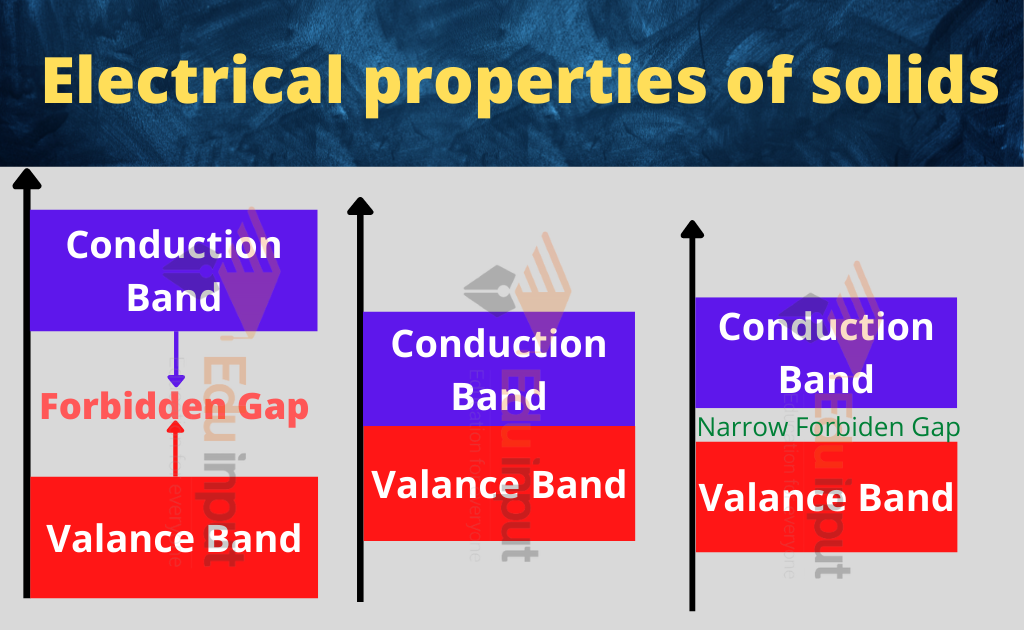
Superconductors are those materials that have zero resistance against the current.
Magnetic Properties
The magnetic properties of solids can be explained by domain theory. Depending upon the net magnetic effect in atoms the substances are classified into three types.
Solids can be classified by their hysteresis loop.
Types of Solids
These properties depend upon the type of solid, and solids can be broadly classified into the following categories:
- Crystalline Solids
- Amorphous solids
- Polymeric Solids
Amorphous Solids
These solids are without any specific form or structure. In amorphous solids, there is no regular arrangement of the molecules. These solids are also called glassy solids. For example ordinary glass.
Polymeric Solids
They are said to be more or less solid materials because their structure is in between an ordered and a disordered structure. For example nature rubber.
Crystalline solids
Crystalline solids are the third type of solid. Crystalline solids are the hardest and densest type of solids. They are also called crystalline because they form a perfect lattice structure. For example sodium chloride.
Application of Solid-State Physics
Solid-state physics is an important part of our daily lives. The most important applications of solid-state are electronic devices, such as mobiles and computers.
The principles of solid-state physics are the basis of modern technology. Simple things like the wiring in a building or the windowpanes in a house are dependent on the principles derived from solid-state physics.
Related FAQs
Is solid-state physics quantum mechanics?
Physical theories and mathematical derivations that describe the energy at the atomic and subatomic levels are dealt with in the laws of quantum mechanics. The basic physical laws of Solids and their related properties are more related to solid-state physics. Both branches of physics are related to one another but are used separately.
In which state do ionic compounds conduct electricity?
It is possible for the ionic compounds to conduct electricity if they are free to move. That is the reason why ionic compounds act as conductors when dissolved in water.
What do you mean by solid-state?
A solid-state is a state of matter in which particles are fixed. They can not move freely and they retain their boundaries without support.
What is a lattice point in solid-state physics?
When an amorphous structure is cooled slowly or gradually at a constant temperature, it eventually crystallizes into a periodic structure. These lattice points represent the position of the atoms in a crystal.





Leave a Reply