Sp2-hybridization, definition, explanation, examples and significance
Definition
The process in which one s and two p orbitals intermix to form three sp2 –hybrid orbitals is called sp2 –hybridization.
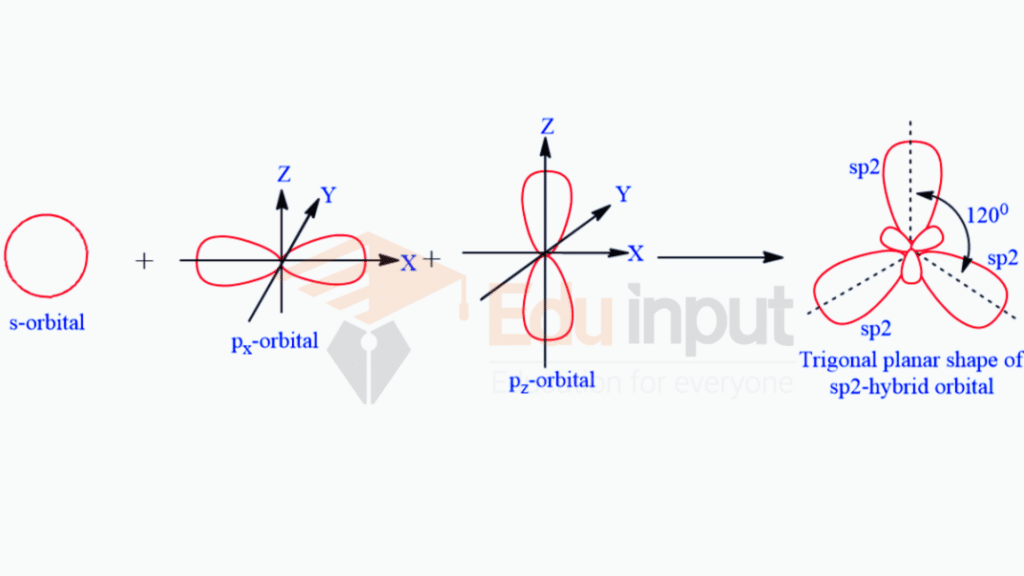
Explanation
sp² hybridization stands as a crucial concept, defining the geometry and bonding characteristics of numerous compounds. This hybridization involves the mixing of one s orbital and two p orbitals, resulting in three equivalent sp² hybrid orbitals. The outcomes of sp² hybridization play a pivotal role in shaping molecules with trigonal planar geometry.
sp² hybridization is a basic concept in chemistry that governs the structural and reactivity aspects of various examples of chemical compounds. Its impact on molecular geometry, conjugation in organic compounds, and contributions to the electronic properties of materials make it a pivotal topic in the study of chemical science. Understanding sp² hybridization opens doors to exploring the intricacies of molecular architecture and lays the foundation for advancements in various chemical disciplines.
sp2-hybridization Examples
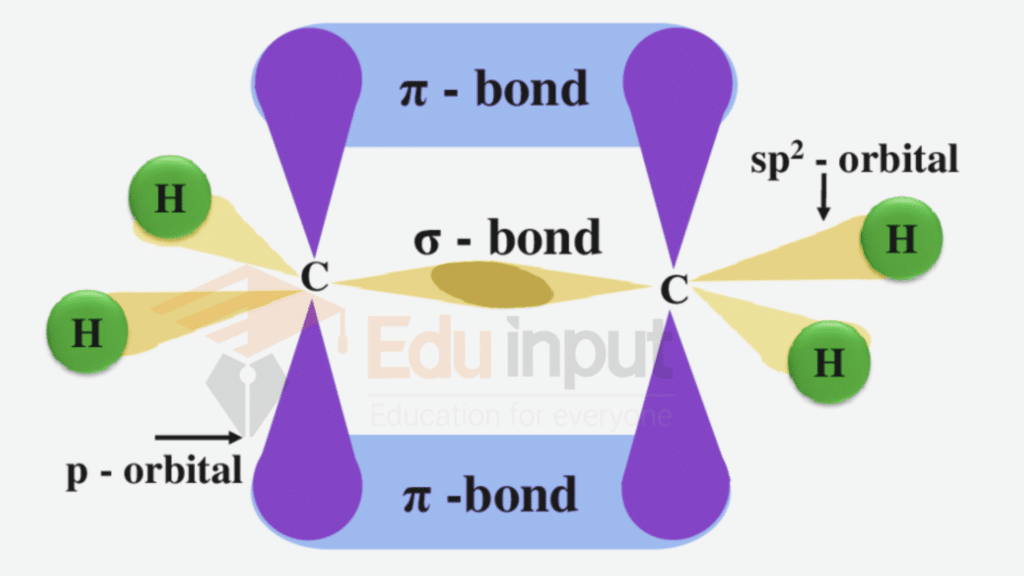
Ethene C2H4
The electronic configuration of the valence shell of C(6):

In the formation of ethene, the carbon atom undergoes sp²-hybridization to give three sp2– hybrid orbitals which are coplanar and oriented at an angle of 120°. One of the p orbitals (pz) of each carbon remains hybridized which is perpendicular to the planar hybrid orbitals
Among three sp2– hybrid orbitals, one sp²-hybrid orbital overlaps with the sp²-hybrid orbital of other carbon atoms to form a C – C sigma bond. The other two sp²-hybrid orbitals of each carbon atom overlap with the 1s orbital of two hydrogen atoms. Hence, in an ethene molecule, four sigma bonds are formed due to sp2-s (C – H) overlapping and one sigma bond due to sp2– sp2 overlap (C-C). The hybridized partially filled p orbitals undergo sideways overlap to form a π-bond.
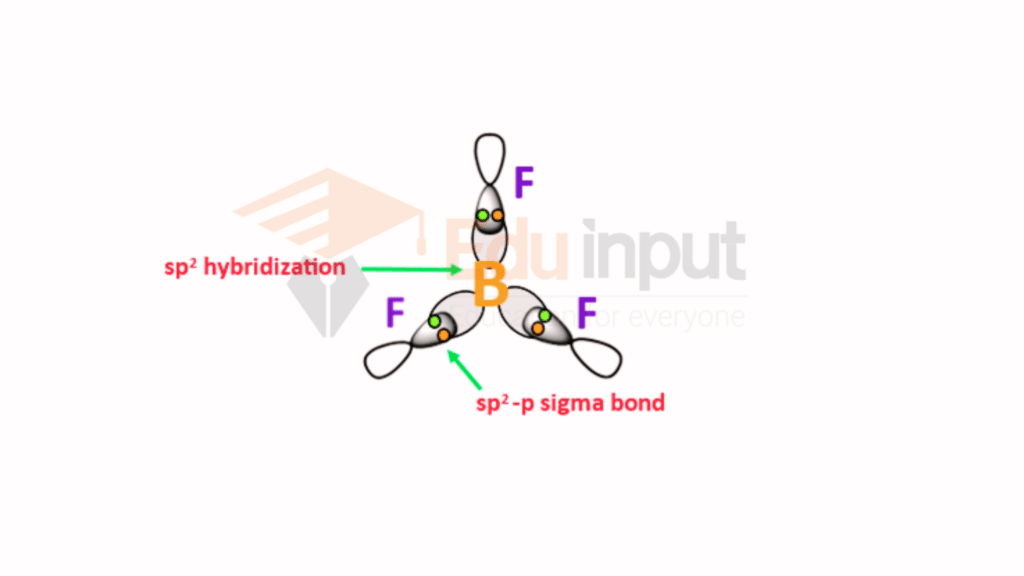
BF3
The electronic configuration of the valence shell of B(5).

In the ground state, boron has only one unpaired electron. In the excited state, one electron from 2s is promoted to 2py. Hence, boron becomes trivalent. Now one 2s and two 2p (2px and 2pz) orbitals hybridize to give three sp2-hybrid orbitals.
These three sp2 hybrid orbitals are half-filled planar and oriented at the angle of 120. Electronic configuration of the valence shell of F(9).
In fluorine, one of the p orbitals is half-filled. In BF3, three sp2-hybrid orbitals of boron overlap with p orbitals of three fluorine atoms.
Significance of sp² Hybridization
1. Conjugated Systems in Organic Chemistry
Application: sp² hybridization is fundamental in understanding and predicting the behavior of conjugated systems, such as those found in aromatic compounds and molecules with alternating single and double bonds.
2. Pi (π) Bond Formation
Role in Double Bond Formation: The unhybridized p orbital in sp² hybridization enables the formation of pi (π) bonds. In molecules with double bonds, the unhybridized p orbitals overlap to create π bonds, contributing to the overall stability.
3. Planar Molecular Structures
Impact on Geometry: The trigonal planar geometry resulting from sp² hybridization is a key factor in determining the planar structures of molecules. This geometry affects the physical and chemical properties of the compounds.
4. Electronic Properties of Materials
Influence on Conductivity: In materials science, the sp² hybridization of carbon atoms in graphene imparts unique electronic properties, contributing to the material’s exceptional electrical conductivity.
5. Biological Molecules
Role in Amino Acids and Proteins: sp² hybridization is observed in the amino acid glycine and other amino acids, influencing the planar structures of peptide bonds and contributing to the folding and stability of protein structures.
6. Advanced Organic Synthesis
Use in Organic Reactions: Understanding sp² hybridization is crucial in planning and executing organic synthesis. It guides chemists in predicting reaction pathways and designing efficient synthetic routes.
7. Catalysis and Chemical Reactivity
Influence on Catalytic Processes: The planar geometry and reactivity associated with sp² hybridization play a role in catalytic processes, influencing reaction mechanisms and selectivity in various chemical transformations.
What is sp² hybridization?
sp² hybridization is a type of hybridization in which one s orbital and two p orbitals of an atom combine to form three sp² hybrid orbitals. These orbitals have a trigonal planar arrangement around the central atom.
Which atoms commonly undergo sp² hybridization?
Atoms like carbon and nitrogen commonly undergo sp² hybridization. It is frequently observed in molecules with double bonds or in compounds with a trigonal planar geometry.
What is the significance of sp² hybridization in molecular geometry?
sp² hybridization results in a trigonal planar molecular geometry. This arrangement is crucial in determining the overall shape of molecules, affecting their physical and chemical properties.
How many sigma (σ) bonds are formed in sp² hybridization?
In sp² hybridization, each hybrid orbital forms one sigma (σ) bond. Therefore, if an atom undergoes sp² hybridization and is involved in three sigma bonds, it will have three sp² hybrid orbitals.
How does sp² hybridization contribute to pi (π) bond formation?
The unhybridized p orbital resulting from sp² hybridization is available for pi (π) bond formation. In molecules with double bonds, these unhybridized p orbitals overlap to create π bonds.





Leave a Reply