Radioactivity- Definition and Types of Radioactivity
Radioactivity is a process in which radiations are produced spontaneously. Radiations are emitted from radioactive elements and the phenomenon is called radioactivity.
Radioactivity
It has been observed that those elements whose charge number Z is greater than 82 are unstable. Some invisible radiations, that can affect the photographic plates emanate out of these elements. Such elements are called radioactive and the phenomenon is called radioactivity.
The radiation coming out of the radioactive elements is called alpha, beta, and gamma radiation. The interaction of radiation with matter produces ionization in that matter.
Radioactivity is purely a nuclear phenomenon. Whenever any radiation is emitted out of any radioactive element it is always accompanied by some changes. This phenomenon is called radioactive decay or nuclear transmutation.
These radiations can be detected with radiation detectors. Geiger Muller counter and Wilson Cloud Chamber are types of radiation detectors.
Uses of these radiations in daily life have increased a lot. For example, nuclear reactors produce the electric current by fission reaction of these radioactive elements. There are also biological effects of radiation.
Discovery of Radioactivity
Radioactivity was discovered by Henri Becquerel in 1896 He found that an ore containing uranium (Z= 92) emits invisible radiation that penetrates through a black paper wrapping a photographic plate and affects the plate.
After Becquerel’s discovery, Marie Curie and Pierre Curie discovered two new radioactive elements they called polonium and radium.
Experiment for the discovery of Radioactivity
The analysis of the radiations radiating out of radioactive material can be carried out by a simple experiment.
The radioactive material is placed at the center of a block of lead by drilling a hole in the block. Radioactive radiations enter a vacuum chamber after emerging out of this hole.
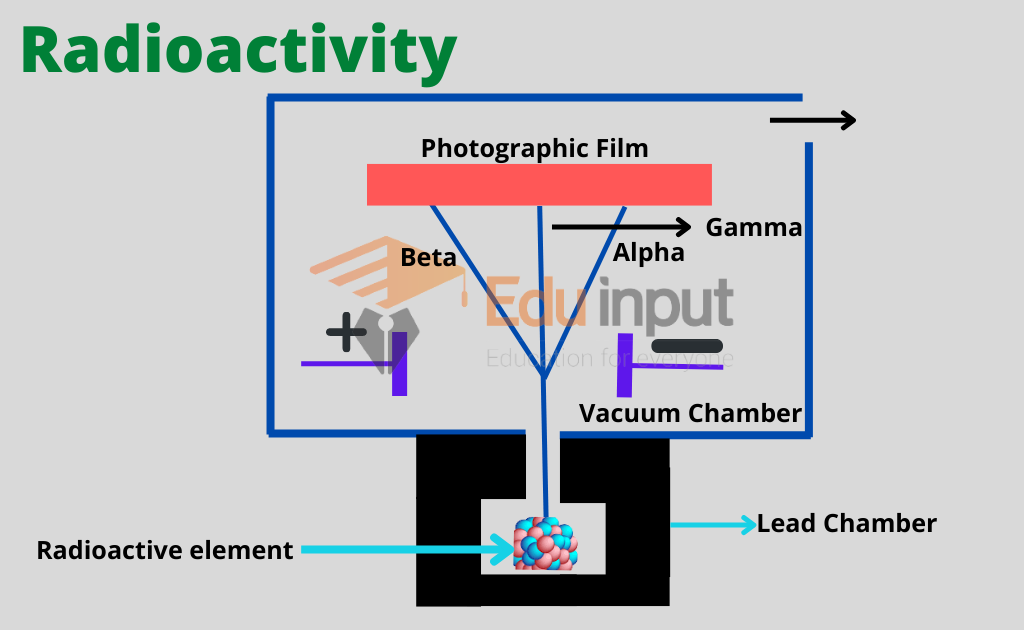
After passing between the two parallel plates the radiations strike a photographic plate. These radiations, instead of impinging at one point, fall at three different points due to the potential difference between the plates.
Types of Radiation
Three types of radiation are emitted by naturally occurring radioactive elements.
From this experiment, it can be concluded that all radiations from the radioactive material are not alike.
The radiation that bends towards the negative plate is made up of positively charged particles. These are called α-particles.
Those radiations that bend towards the positive plate are composed of negatively charged particles. These are called β-particles.
Those radiations that go straight without bending have no charge on them. These are called γ-rays.
Further experiments expose that
- α-particles are helium nuclei. The charge on them is +2e while their mass is 4u (atomic mass unit) that is every α-particle has two protons and two neutrons in it.
- β-particles are in fact fast-moving electrons that come out of the nucleus of a radioactive element.
- γ-rays like X-rays are electromagnetic waves that issue out of the nucleus of a radioactive element. The wavelength of these rays is much shorter, compared to the wavelength of X-rays.






Leave a Reply