4 DEFECTS OF BOHR’S ATOMIC MODEL | Limitations of Bohr’s Atomic Model
DEFECTS OF BOHR’S ATOMIC MODEL
There are so far four major defects in the atomic model of Bohr.
1. Multi- electron system can’t be explained by Bohr’s theory. A hydrogen atom has only one electron. Its spectra can be explained by Bohr’s model.
But the spectra of He, Li, and Be which have 2, 3, and 4 electrons respectively can’t be explained. Anyhow, the spectra of He+, Li2+, and Be3+ can be explained, because they have only one electron like hydrogen atoms.
2. The fine structure of hydrogen atoms can’t be explained by Bohr’s theory. When the spectroscopes of high resolving power are used to study the spectra of hydrogen atoms, then some weak lines are also obtained.
These fine lines lie very close to each other. For example, the Hα line in the Balmer series is found to consist of five component lines. It means that in a given orbit (shell or energy level), there are several orbitals. But Bohr did not know that.
3. Bohr proposed that electrons move in a circular orbit around the nucleus and the orbit is planar. These days, we are convinced that the motion of the electron is not limited to a single plane, but takes place in three-dimensional space.
4. Explanation of the Zeeman effect and Stark effect are not available from Bohr’s model.
(a) Zeeman Effect
When an excited atom that gives a line emission spectrum is put into the magnetic field, its spectral lines are spilled up into still thinner. These lines are closely spaced. This splitting is called the Zeeman effect.
(b) Stark effect
When the spectral lines are split up into a number of thinner lines by applying the electrical field, then it is called the Stark effect.
Also read
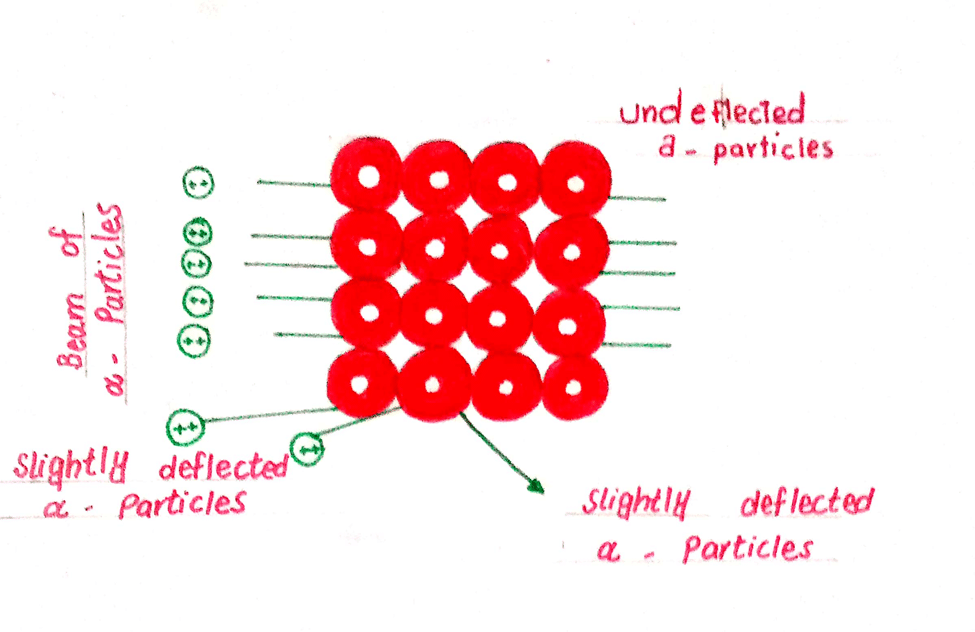
- RUTHERFORD’S ATOMIC MODEL (DISCOVERY OF NUCLEUS)
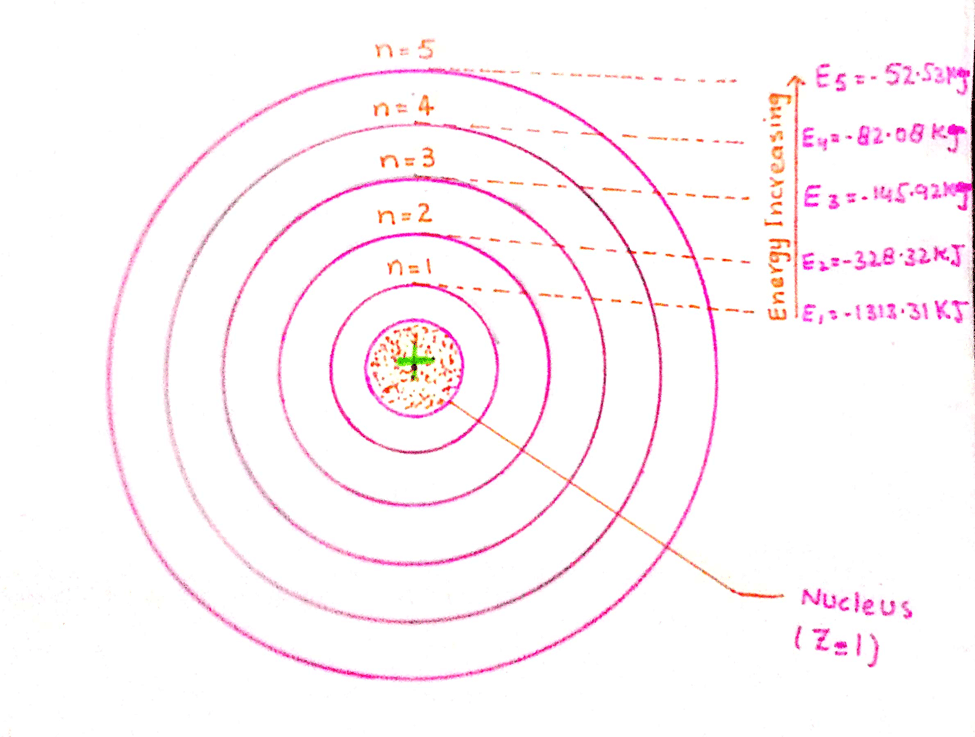
- What is an atomic model in chemistry?
- Planck’s Quantum Theory

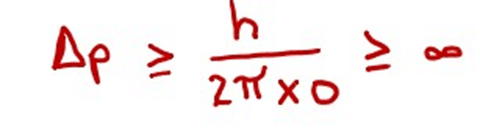


Leave a Reply