Ionic solids-types, formation, properties, crystal structure and uses
What are ionic solids?
Ionic solids are a type of crystalline solid in which the constituent particles are ions—electrically charged atoms or groups of atoms.
Ionic solid definition
These solids are held together by ionic bonds, which are electrostatic attractions between oppositely charged ions.
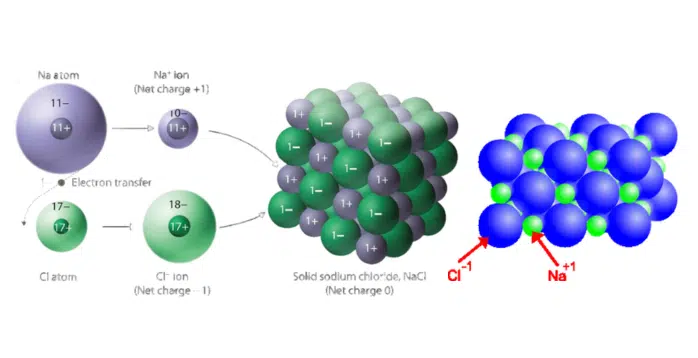
Crystal Structure: Exploring the arrangement of ions in ionic solids
The crystal structure of ionic solids refers to the arrangement of ions within the solid lattice. Ions are atoms or groups of atoms that have gained or lost electrons, resulting in a positive or negative charge.

In ionic solids, cations (positively charged ions) and anions (negatively charged ions) are held together by strong electrostatic forces, forming a three-dimensional lattice structure. The arrangement of ions in this lattice is determined by the size and charge of the ions, as well as the ratio of cations to anions.
The crystal structure of ionic solids can be classified into different types based on the arrangements of the ions. One common type is the simple cubic structure, where each cation is surrounded by anions and vice versa.
Another type is the body-centered cubic structure, where each cation is surrounded by anions, and there is an additional cation in the center of each cube. There are also more complex crystal structures like the face-centered cubic and hexagonal close-packed structures.
The specific arrangement of ions in a crystal lattice dictates many of the physical and chemical properties of the ionic solid, such as its density, melting point, and conductivity. Understanding the crystal structure of ionic solids can provide insights into their behavior and potential applications.
Formation of ionic solids
Ionic solids are formed through ionic bonding, which involves the transfer of electrons between atoms. In this type of bonding, one atom donates its electrons to another atom, resulting in the formation of positively charged ions (cations) and negatively charged ions (anions). The electronegativity difference between the two atoms determines the strength of the ionic bond. When the electronegativity difference is large, the transfer of electrons is more likely to occur, leading to the formation of a stable ionic solid.
The transfer of electrons in ionic bonding occurs due to the preference of atoms to achieve a stable electron configuration. Typically, metal atoms, which have a lower electronegativity, readily lose their valence electrons to the more electronegative non-metal atoms.
As a result, the metal atom becomes a cation with a positive charge, and the non-metal atom becomes an anion with a negative charge. The attractive forces between the oppositely charged ions then hold the ionic solid together in a three-dimensional lattice structure. This transfer of electrons and the formation of ions is crucial for the formation of ionic solids, which have distinct properties and applications in various fields.
Understanding strong electrostatic forces that hold ionic solids together
The strength of the electrostatic forces in ionic bonding can be attributed to the large difference in electronegativity between the metals and non-metals involved. In the case of ionic bonding, the non-metal has a higher electronegativity than the metal, causing it to attract and hold onto the electrons transferred from the metal. This strong attraction between the oppositely charged ions ensures the stability of the ionic solid. It is this strong electrostatic force that gives rise to the unique physical and chemical properties exhibited by ionic solids.
Properties of ionic solids
Ionic solids possess a range of unique physical properties that set them apart from other types of solids.
1. High Melting and Boiling Points
- Attributed to strong electrostatic forces between ions.
- Magnitude depends on ion charge and size.
- Breaking crystal lattice structure demands substantial energy.
- Resulting in exceptionally high melting and boiling points.
2. Brittle Nature
- Crystalline structure makes ionic solids often brittle.
- Application of force induces slight shifts in ion layers.
- Rigidity and strong electrostatic forces resist shifts, causing fractures.
- Regular ion arrangement creates planes of weakness, facilitating breakage.
3. Electrostatic Forces of Attraction
- Primary force binding ions in crystal lattice.
- Arises from attraction between oppositely charged ions.
- Determines the overall stability of the ionic solid structure.
4. Crystal Lattice Structure
- A three-dimensional, ordered arrangement of ions.
- Maximizes attractive forces between ions.
- A key factor in the unique properties of ionic solids.
5. Dependency on Ion Properties
- Physical properties are influenced by ion charge and size.
- Larger charges and smaller sizes contribute to stronger electrostatic forces.
- Reflects in the overall behavior and characteristics of the ionic solid.
6. Energy Requirements for Structure Breakage
- Substantial energy is needed to disrupt the crystal lattice.
- Relates to the formidable electrostatic forces holding ions together.
- High energy input is required for melting or boiling ionic solids.
7. Formation of Planes of Weakness
- Regular ion arrangement creates specific orientations.
- Leads to the development of planes of weakness in the crystal lattice.
- Facilitates controlled fracture along these planes, contributing to brittleness.
Energy required to break apart an ionic solid into its constituent ions
Lattice energy refers to the amount of energy needed to separate an ionic solid into its ions in the gas phase. This energy is a measure of the strength of the electrostatic forces holding the ions together in the crystal lattice. In simple terms, it represents the energy required to overcome the attraction between positively and negatively charged ions and convert them into independent, gaseous ions.
The lattice energy is influenced by various factors, including the charge of the ions and the distance between them. As the magnitude of the charges increases, the lattice energy also increases because the electrostatic forces become stronger. Additionally, as the distance between the ions decreases, the lattice energy increases due to the closer proximity of the charges. Thus, compounds with higher charges and smaller ion sizes tend to have higher lattice energies. Understanding the lattice energy of an ionic solid is crucial in predicting its stability and reactivity, as well as determining its physical properties such as melting and boiling points.
Solubility of ionic solids in different solvents
Ionic solids, due to their unique crystal structure and strong electrostatic forces, display varying degrees of solubility in different solvents. Solubility refers to the ability of a solute— in this case, an ionic solid—to dissolve in a given solvent, resulting in a homogeneous mixture known as a solution.
The solubility of ionic solids is influenced by several factors, including the nature of the ions involved, the polarity of the solvent, and the temperature at which the dissolution occurs.
The solubility of ionic solids is primarily determined by the balance between the attractive forces holding the ions together in the solid lattice and the attractive forces between the ions and the solvent molecules.
In general, polar solvents, such as water, tend to dissolve ionic solids more readily than nonpolar solvents, due to their ability to interact with the charged ions through dipole-dipole interactions. This is because the polar solvent molecules are capable of effectively solvating the ions, surrounding and stabilizing them in solution.
Conversely, nonpolar solvents, such as hydrocarbons, have limited interactions with ions and therefore exhibit lower solubility for most ionic solids. Temperature also plays a role in solubility, with higher temperatures generally increasing solubility as the kinetic energy of the particles increases, allowing for more frequent and successful collisions between solute and solvent molecules.
Types of Ionic Solids
Ionic solids are a broad category of compounds that are formed through the transfer of electrons between a metal and a non-metal. These compounds are characterized by the strong electrostatic forces that hold the positively and negatively charged ions together in a regular arrangement. The different types of ionic solids can be classified based on the specific arrangements of ions within their crystal structures, which greatly influence their properties and behaviors.
One common type of ionic solid is the simple cubic structure, where each cation is surrounded by anions and vice versa. This arrangement results in a repeating pattern where ions occupy the corners of each cube.
Another type is the body-centered cubic structure, where in addition to the corner ions, there is an additional ion present at the center of each cube. This arrangement creates a more closely packed structure with higher coordination numbers for the ions. A third type is the face-centered cubic structure, where ions are located at the corners and faces of each cube. This arrangement is the most closely packed and results in the highest density among the different types of ionic solids.
These various arrangements of ions in ionic solids give rise to different physical and chemical properties. The packing of ions in the crystal lattice influences the melting and boiling points, as well as the hardness and brittleness of the solid. Additionally, the arrangement of ions affects the conductivity of the solid, as the movement of ions within the lattice is responsible for the flow of electric current.
Understanding the different types of ionic solids and their ion arrangements is crucial in predicting and explaining the behavior of different compounds in various applications, such as in battery materials, ceramics, and pharmaceuticals.
Applications of ionic solids
Ionic solids find wide-ranging applications in various industries and technologies due to their unique properties.
| Application | Ionic Solid | Description |
| 1. Electrolytes | Sodium chloride (NaCl) | Ionic solids such as NaCl are used as electrolytes in batteries and fuel cells, facilitating the conduction of ions between electrodes, enabling the flow of electric current. |
| 2. Building Materials | Calcium carbonate (CaCO3) | Ionic solids like CaCO3 (calcite) are used in the construction industry for making cement and concrete. These materials provide strength and durability to structures. |
| 3. Pharmaceuticals | Ionic solids such as NaCl are used as electrolytes in batteries and fuel cells, facilitating the conduction of ions between electrodes, and enabling the flow of electric current. | Ionic solids are used in antacids and laxatives. Mg(OH)2, for example, is used to neutralize excess stomach acid and alleviate indigestion. |
| 4. Food Additives | Sodium chloride (NaCl) | NaCl is commonly used as table salt in food, providing flavor enhancement. Other ionic compounds, such as potassium chloride, are also used as salt substitutes. |
| 5. Metallurgy | Aluminum oxide (Al2O3) | Ionic solids like Al2O3 are used as a flux in metallurgical processes to remove impurities during the extraction of metals from ores, improving the efficiency of metal refining. |
| 6. Pharmaceuticals | Lithium carbonate (Li2CO3) | Ionic solids are used in psychiatric medications, such as lithium carbonate, to treat bipolar disorder and stabilize mood. |
| 7. Glass Manufacturing | Sodium carbonate (Na2CO3) | Ionic compounds like Na2CO3 are used in the manufacturing of glass. They act as fluxing agents, reducing the melting point of silica and aiding in the glass-forming process. |
| 8. Water Softening | Calcium sulfate (CaSO4) | Ionic solids like CaSO4 are used in water softeners to remove excess calcium and magnesium ions, preventing the buildup of scale in pipes and appliances. |
| 9. Photocatalysis | Titanium dioxide (TiO2) | TiO2, an ionic solid, is used in photocatalytic processes, such as in self-cleaning surfaces and air purification, where it reacts with light to catalyze chemical reactions. |
| 10. Piezoelectric Devices | Lead zirconate titanate (Pb(Zr,Ti)O3) | Ionic solids with piezoelectric properties, like PZT, are used in sensors, actuators, and devices converting mechanical energy into electrical signals or vice versa. |






Leave a Reply