Atomic radius-Definition, examples, variation in periodic table
What is Atomic Radius?
Atomic radius is the distance from the center of an atom's nucleus to its outermost electron.
Example
Atomic radius of Na = 154 pm
Atomic radius of Cl = 99 pm
It is an essential property of an atom and plays a vital role in determining the chemical and physical properties of elements. The size of an atom is determined by the arrangement of its electrons, and the electrons are arranged in energy levels or shells around the nucleus. The size of an atom increases as you move down a group in the periodic table and decreases as you move from left to right across a period.
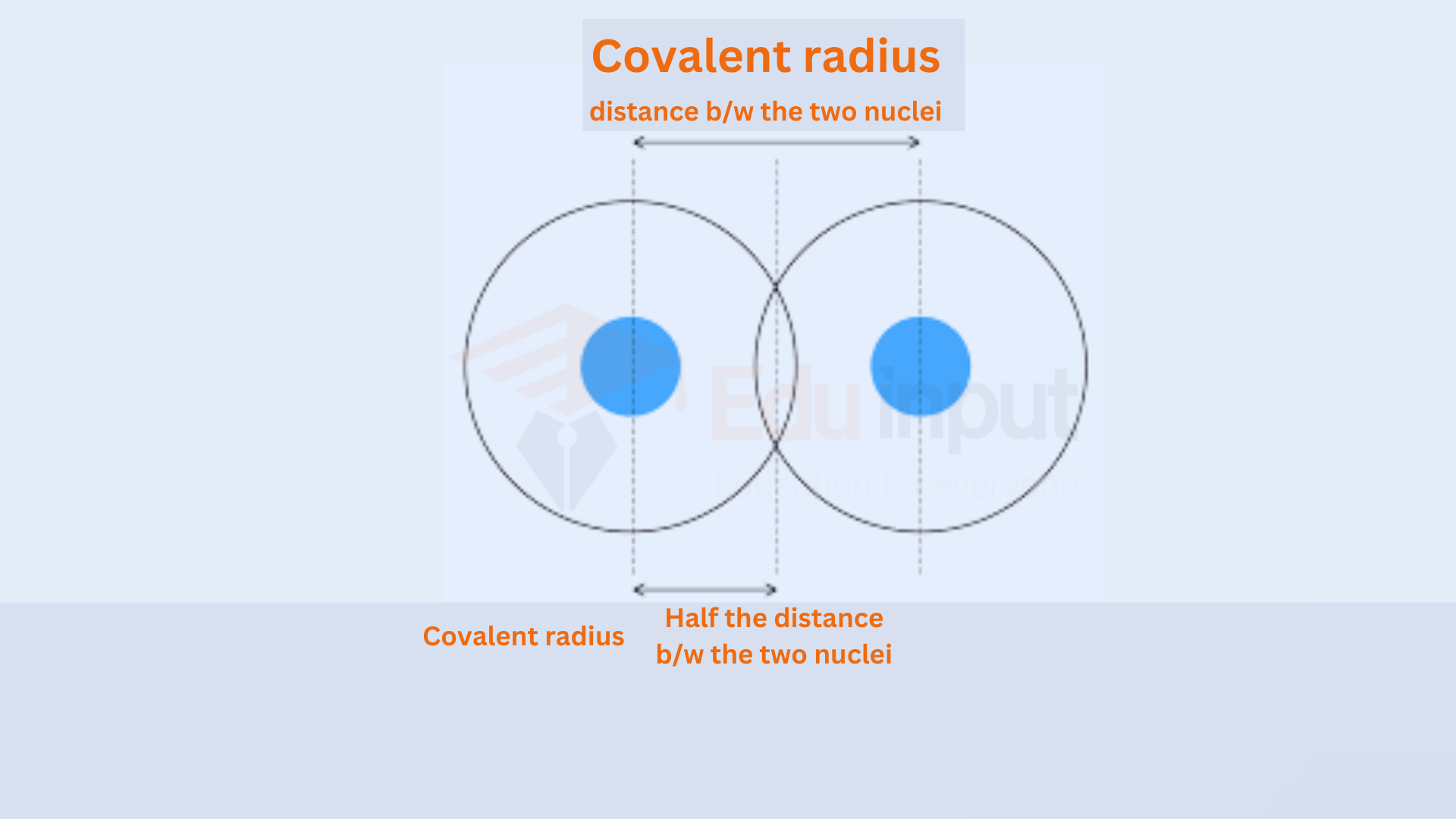
How is Atomic Radius Measured?
Several methods can be used to measure the atomic radius. X-ray Diffraction involves passing X-rays through a crystal to measure the angle at which they diffract. This angle can be used to determine the distance between the atoms in the crystal.
The atomic force microscope is used for measuring atomic radius. It uses a probe to measure the force between the probe and the atoms on the surface.
The radius of an Atom cannot be found accurately:
The radius of an atom cannot be determined precisely due to the following reasons.
1. Atom has no definite/sharp boundary.
2. The electronic probability distribution is affected by its neighboring atoms. 3. Atomic radius changes from compound to compound
Periodic Trends of atomic radii
Atomic radius variation can be observed across periods, groups, and transition metals.
Variation of atomic radii in period
In periods, atomic radii generally decrease from left to right because electrons are added to the same shell, resulting in an increase in effective nuclear charge and a decrease in atomic size. The screening effect remains constant across the period. For example, in the second period, the atomic radius of lithium (Li) is the greatest, whereas neon (Ne) has the smallest atomic radius.
Variation of atomic radii in period
Atomic radii generally increase from top to bottom in groups because a new shell is added with each successive element and the shielding effect increases. The increase in shielding effect reduces the nuclear pull for valence electrons, increasing atomic size. For instance, in group I-A, lithium (Li) has the smallest atomic radius, while francium (Fr) has the largest.
Variation of atomic radii in transition metals
When it comes to transition metals, the atomic radius variation is not as straightforward. As we move from left to right in transition elements Sc (21)-Zn(30), Y(39)-Cd(48), intervening electrons come into play, and the screening effect (also known as the shielding effect) reduces the force of attraction between the nucleus and the valence electrons. This results in a decrease in the atomic size.
Importance of atomic radius
Atomic radius is an important property of atoms. It affects chemical and physical properties such as melting temperature and density. For example, the atomic radius of a chemical element affects its reactivity.
Bigger atomic radii make elements more reactive, so elements with big atoms tend to be more reactive.
In contrast, elements with smaller atomic radii tend to be less reactive because the outer electrons of the element have a closer distance to the nucleus, thus, making the atom less reactive.
Applications of atomic radius
There are several practical applications of atomic radius in various fields. It is used to determine the properties of materials and to design new materials with specific properties In the field of catalysis, atomic radius is used to design catalysts with certain shapes and sizes.
FAQs
Q: How does atomic radius relate to the periodic table?
Atomic radius generally increases as you move down a group in the periodic table and decreases from left to right across a period. For detailed calculations and insights, try the Periodic Table Calculator.
Q: Can atomic radius be negative?
A: No, atomic radius cannot be negative. It is a measure of the distance from the center of the nucleus to the outermost electron.
How is atomic radius related to the shielding effect?
A: The shielding effect occurs when the electrons in the inner energy levels shield the outer electrons from the pull of the positively charged nucleus. This results in a larger atomic radius.
What is the difference between atomic radius and ionic radius?
A: Atomic radius refers to the distance from the center of an atom’s nucleus to its outermost electron, while ionic radius refers to the distance between the center of an ion’s nucleus and its outermost electron.
What are some practical applications of atomic radius?
A: Atomic radius has various practical applications, such as in materials science and catalysis, where it is used to design materials and catalysts with specific properties.






Leave a Reply