Which Properties of Solvents are Useful for Solvent Extraction?
Some properties that are useful for solvent extraction are solvent selectivity, its immiscibility with water, boiling point, viscosity, chemical reactivity, density, and flash point.
Also, Read: Factors That Affect Solvent Extraction
Properties of Solvents for Efficient Solvent Extraction
Here are the Properties of Solvents that are needed for Efficient Solvent Extraction:
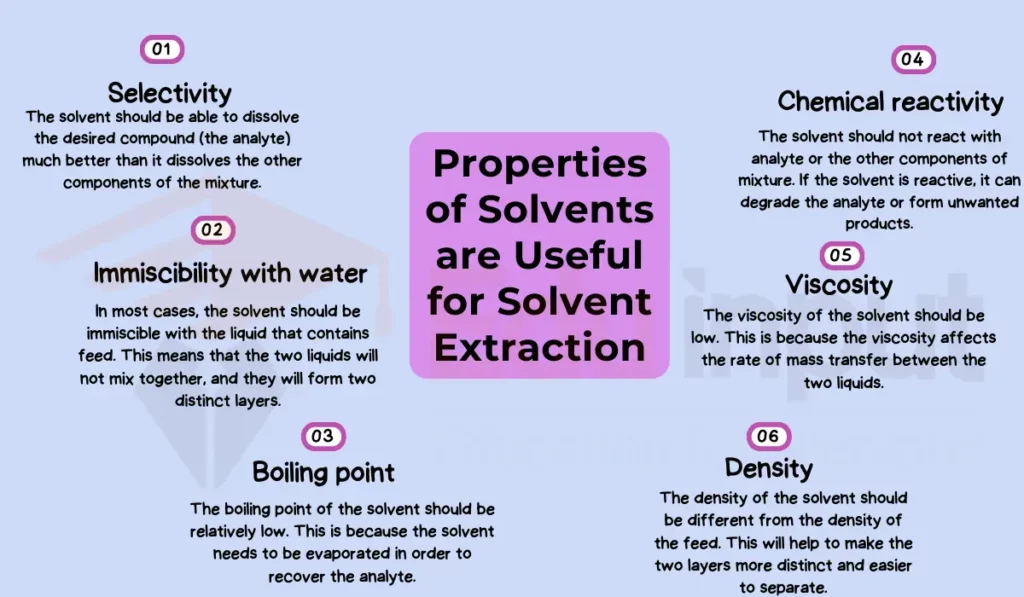
Selectivity
The selectivity of a solvent refers to its ability to preferentially dissolve the target compound over other components in a mixture. This property relies heavily on the chemical affinity between the solvent and respective solutes.
A highly selective solvent will isolate the desired compound while leaving behind unextracted materials. Chloroform shows strong selectivity for caffeine extraction from coffee beans, attributed to intermolecular interactions between chloroform and caffeine.
Immiscibility with Water
Immiscibility is the inability of a solvent to mix uniformly with water, instead separating into distinct solvent-rich and water-rich phases. This property of solvent facilitates the extraction of organic compounds from aqueous mixtures.
It simplifies the isolation of the solvent phase containing the dissolved product. Due to its immiscibility with water, dichloromethane is commonly employed to extract non-polar lipids from biological fluids.
Boiling Point
A solvent’s boiling point directly impacts the ease of post-extraction processing. Solvents with low boiling points, like hexane, can be easily removed via evaporation, leaving behind extracted material with no residual solvent. Hexane’s low boiling point enables efficient concentration of fragile natural products like essential oils and fragrances.
Chemical Reactivity
Reactivity considers how chemically inert a solvent is under extraction conditions, minimizing unwanted reactions with the solutes of interest. Reactions lead to losses in extract purity and quality. For example, ethanol shows excellent stability with sensitive cannabinoids during cannabis extractions to preserve medicinal properties.
Viscosity
Viscosity quantifies a liquid’s thickness and flow characteristics. In large-scale extraction processes, low-viscosity solvents like toluene ensure adequate contact between solvent and feed material. This facilitates rapid mass transfer, improving extraction efficiency.
Density
A solvent’s density affects phase separation steps post-extraction. The organic phase must have a higher or lower density than the aqueous phase. In bitumen production from oil sands, toluene’s density causes it to sink below water layers, benefiting subsequent solvent recovery.
Flash Point
The flash point measures flammability levels. Depending on temperature settings, a higher flash point might be required for safe operation, especially in large, industrialized extraction equipment. Due to relatively high flash points, kerosene has found utility in supplemental oil extraction from depleted reservoirs.
Toxicity
Toxicity levels require evaluation as more stringent environmental regulations are enacted. Bio-based, greener solvents are being assessed to replace conventional options. Limonene obtained from orange peels demonstrates potential, with low toxicity and strength in dissolving hydrophobic compounds.





Leave a Reply