Why is Diffusion Faster in Hot Water?
Increasing the temperature of water causes diffusion to occur more rapidly because heating impacts water at the molecular level. Elevated temperature speeds up diffusion in water through effects on viscosity, hydrogen bonding, solubility, and kinetic energy.

Reasons Why is Diffusion Faster in Hot Water
Here are a few reasons Why is Diffusion Faster in Hot Water:
1. Increased Kinetic Energy
Heating water increases the kinetic energy and motion of its molecules. The faster vibration and collision of the water molecules transfer energy to diffusing particles, speeding up their diffusion.
2. Lower Viscosity
Hot water has a lower viscosity than cold water. This means it flows more easily, providing less resistance to particles moving through it. Lower viscosity enables faster diffusion.
3. Disrupted Hydrogen Bonds
Heated water molecules have more energy. It causes more disruption of hydrogen bonds between them. This makes it easier for particles to maneuver through the liquid and diffuse.
4. Increased Solubility
Some diffusing substances become more soluble in hotter water. Increased solubility provides more opportunities for particle-water interactions to facilitate diffusion.
Example
A drop of food coloring will diffuse through hot water noticeably faster than cold water. The increased energy, lower viscosity, and modified bonds promote faster diffusion.
Related Articles:
Why is Diffusion Faster in Small Cells?
Why is Diffusion Faster in Liquids than Solids?
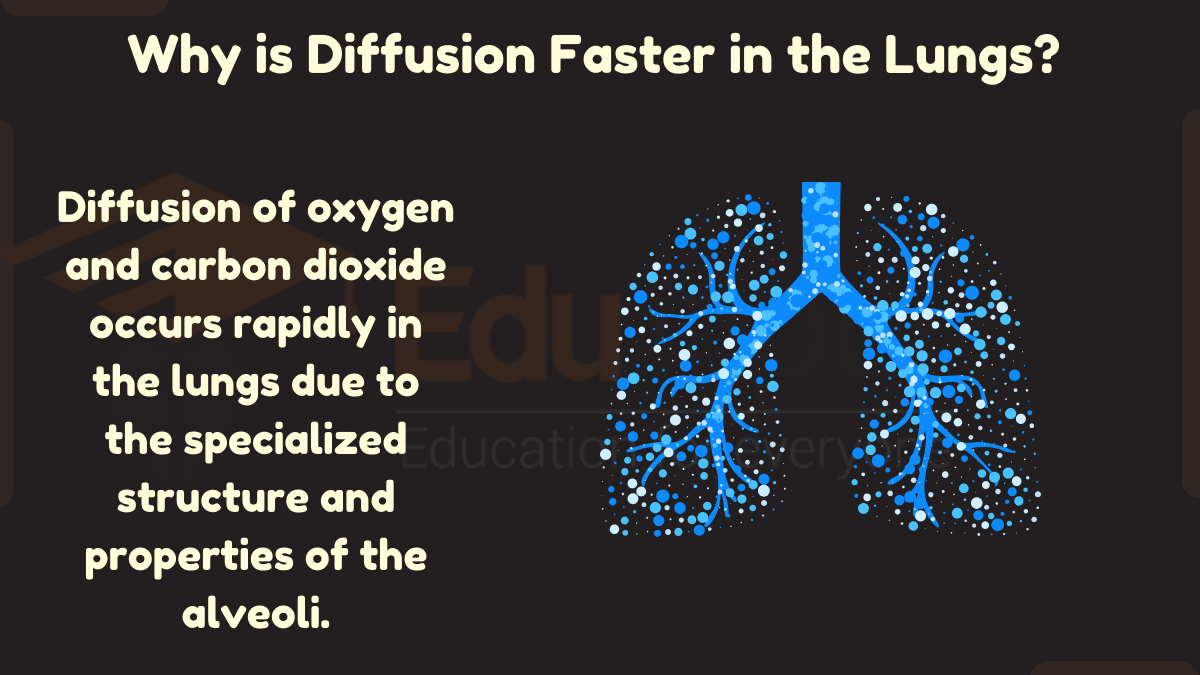
Why is Diffusion Faster in Lungs?
Why is Diffusion Faster in Air Than Water?
Why is Diffusion Faster in Gases?
Why Is Diffusion Faster In Water Than in Agar?



Leave a Reply